 
This means that the negative charge on an electron perfectly balances the positive charge on the proton. Isotopes of an element contain different numbers of neutrons. Each element has its own atomic number, which is equal to the number of protons in its nucleus. Atoms themselves are composed of protons, neutrons, and electrons. Negative and positive charges of equal magnitude cancel each other out. Chemistry is based on the modern atomic theory, which states that all matter is composed of atoms. Neutral atoms have the same number of electrons as they have protons, so their overall charge is zero. We will discuss isotopes and their symbols more in section 4.5 of this chapter. These ancient philosophers speculated that the earth was made up of different combinations of basic substances, or elements. It must be added, atomism was one of a number of competing theories on the nature of matter. 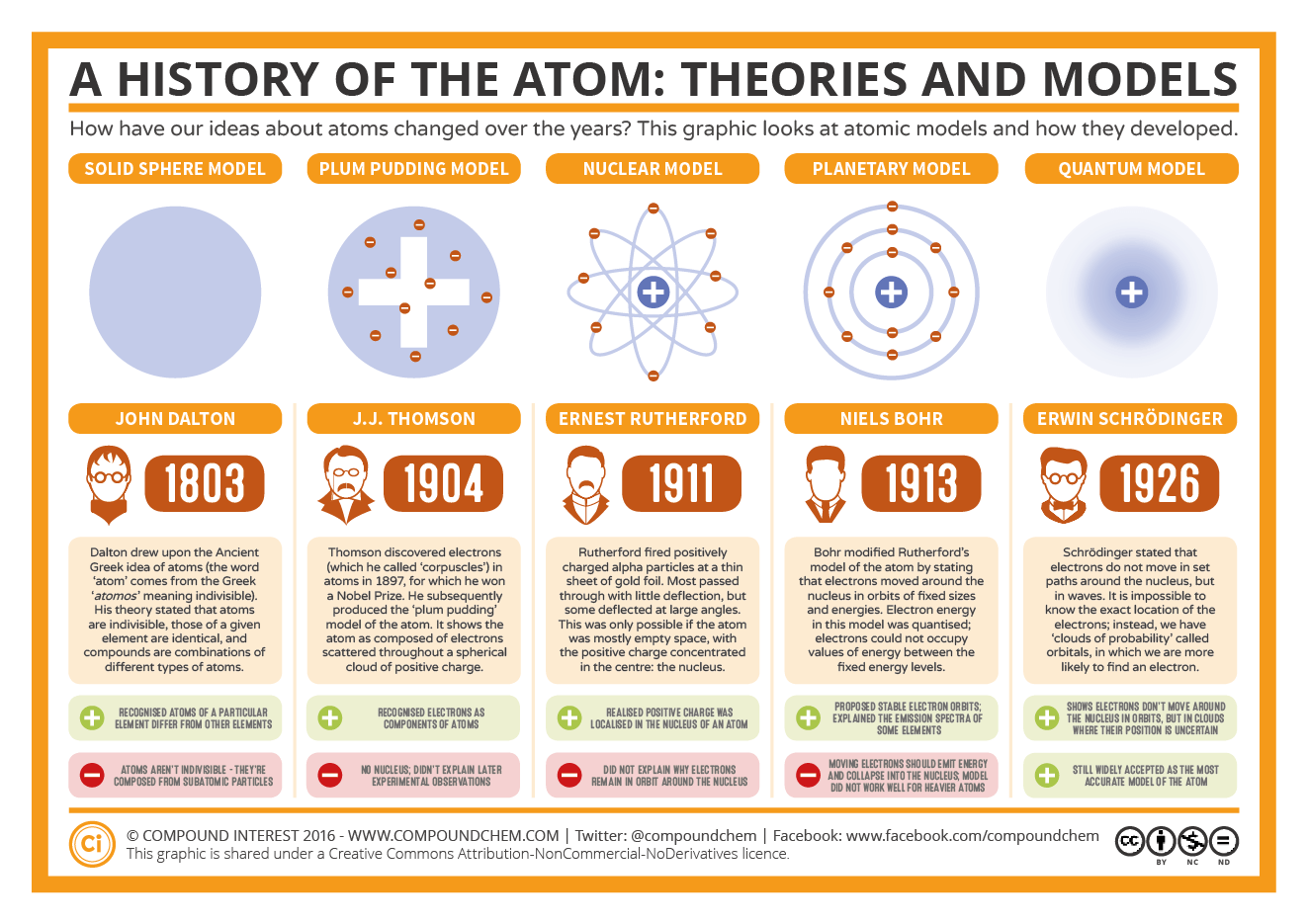
Atoms of the same element (i.e., atoms with the same number of protons) with different numbers of neutrons are called isotopes. The sum of the number of protons and neutrons in the nucleus is called the mass number of the isotope. Atomic theory began as a philosophical concept in ancient Greece and India. Learn how John Dalton proposed that all matter is made of atoms, which are indivisible and have the same properties. Each element has its own characteristic atomic number.Ītoms of the same element can have different numbers of neutrons, however. Thus, hydrogen has an atomic number of 1, while iron has an atomic number of 26. The number of protons in an atom is the atomic number of the element (Z). Atom, smallest unit into which matter can be divided without the release of electrically charged particles. This number of protons is so important to the identity of an atom that it is called the atomic number. All atoms of hydrogen have one and only one proton in the nucleus all atoms of iron have 26 protons in the nucleus. What makes atoms of different elements different? The fundamental characteristic that all atoms of the same element share is the number of protons. Several scientists working on atomic found that atoms were not the smallest possible particles that made up matter, and that different. By the late 1800’s, John Dalton’s view of as the smallest that made up all had held sway for about 100 years, but that idea was about to be challenged. The modern atomic theory states that atoms of one element are the same, while atoms of different elements are different. This module is an updated version of Atomic Theory I. 
\): The Structure of the Atom.Atoms have protons and neutrons in the center, making the nucleus, while the electrons orbit the nucleus. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
